|
We knowthe physical state of all of the elements in the periodic table.We know the reactivity of the alkali metals with water, increasesas you go down the group. The formula is notshown, that is information we need to recall from memory. The symbol for eachelemetn is included in the periodic table. 
We learned some of theinformation the periodic table reveals to us. The periodic table is a very important tool which contains avery large quantity of information. There are plenty of examples of the elements mentioned above with their conductivity, place, appearance, etc.The periodic table is a very important tool which contains a very large quantity of information. Moreover, it also tells how their physical and chemical properties are dissimilar. These differences between metals, non-metals, metalloids and noble gases study material conclude that metals, non-metals, and metalloids are different elements with different properties and nature. Later in 1894, a Scottish chemist “William Ramsay” joined “Rayleigh” to isolate this gas, and then they discovered a new element known as ‘argon’. But, later, he got to know that it was denser compared to the nitrogen formed by the process of liberating it from its compounds. After a century passed, another physicist,Lord Rayleigh”, isolated a gas from the air that was pure according to his assumption and analysis. In 1785, a chemist and physicist, “Henry Cavendish” noticed that air holds a small proportion of less than a percent of a substance that is less active chemically compared to nitrogen. Furthermore, in chemistry and alchemy, the term noble means the reluctance of the metals like platinum, gold, etc., to conduct the chemical reaction. Therefore, these were then known as inert or rare gases. Group 18.Īfter the discovery and identification of the group members, these gases were thought to be rare and chemically inert at the same time. However, their electronic structure and the assumption that some of them can make compounds resulted in an appropriate designation, i.e. This means that their atoms cannot combine with the atoms of other elements for making chemical compounds. These gases are labelled as group 0 in the periodic table because after these gases were discovered, it was found that they were not able to bond with other atoms. The noble gases don’t have any colour, taste, or odour as well as they are non-flammable gases. Metalloids are shiny as well as dull at the same time.Non-metals are present in s and p blocks.Metals are present in s, p, d, and f blocks. 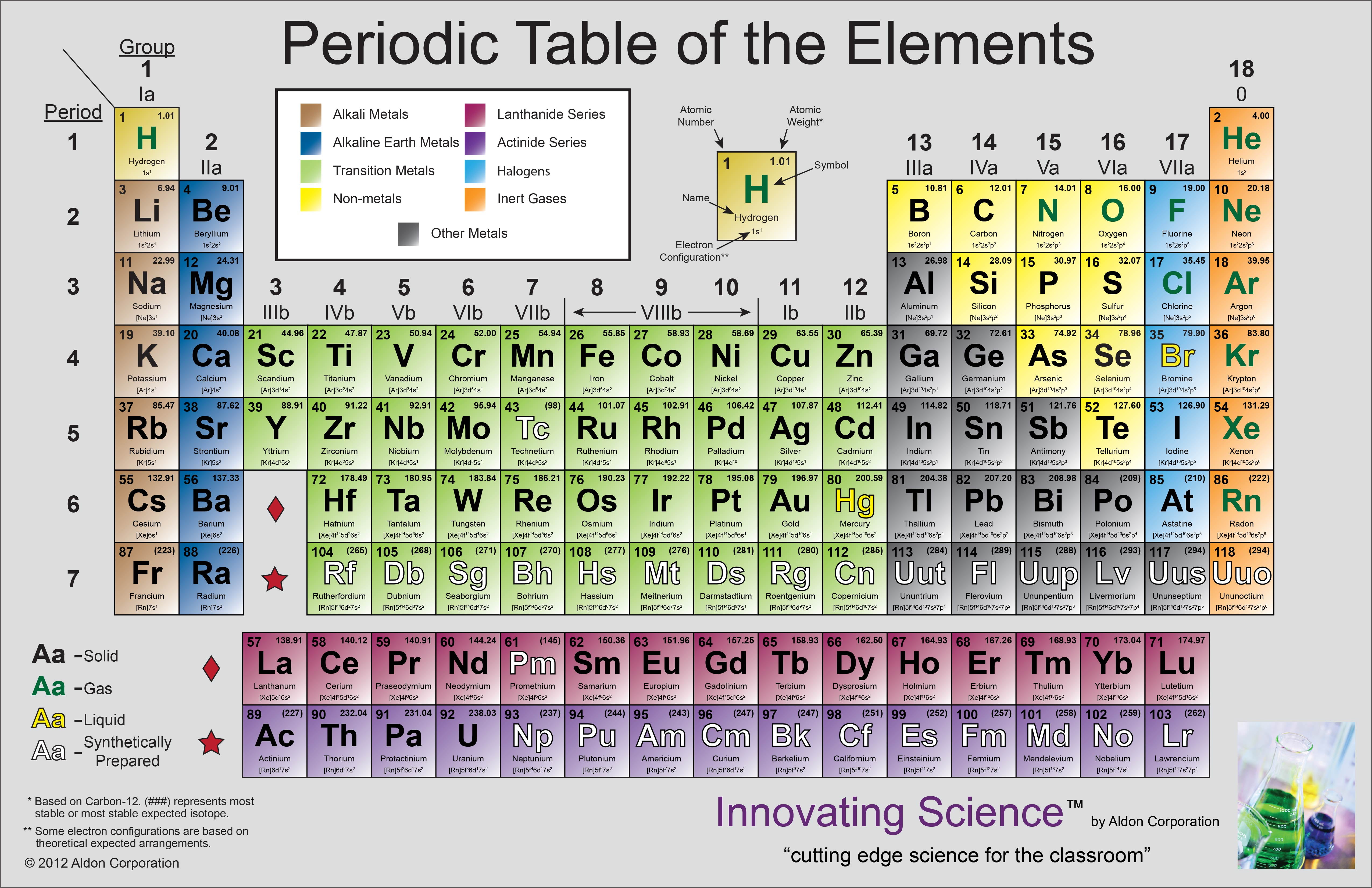
Metalloids are in the centre of the periodic table.Non-metals are on the right in the periodic table.Metals are present on the left side of the periodic table.Metalloids – Tellurium, Polonium, Antimony, Arsenic, etc. Non-metals – Bromine, Iodine, Helium, Argon, Krypton, Neon, Phosphorous, Sulfur, etc. Metals – Sodium, Lithium, Calcium, Barium, Magnesium, Lead, Bismuth, Indium, Iron, Copper, Nickel, Zinc, etc. Moreover, the non-metals are bad conductors of electricity. The metals are good conductors of electricity, metalloids can be a conductor of electricity in some rare cases, and that’s the reason it is also called semiconductors. The metals have less ionisation energy, metalloids have a medium ionisation energy level, while the non-metals are high in ionisation energy. The metals have the highest conductivity of heat, the metalloids have a medium capacity to conduct the heat, and the non-metals are poor when it comes to heat conductivity. Furthermore, the non-metals have high ionisation energy and low conductivity of heat and electricity. Whereas the metalloids fall between the metal and non-metals. 
Metals are generally high in conductivity with less amount of ionisation energy. The difference in Metals, Metalloids, & Non-metals That’s why these are also called semiconductors, and they are also used in computers, electronics, motors, etc. They act more like non-metal in their physical properties, but in some rare cases, they can conduct electricity. Last but not least, metalloids usually have intermediate properties. Moreover, some non-metals are in the gaseous form at room temperature, whereas others are in the liquid or solid-state. They don’t have a ductile or malleable nature. The non-metals are generally bad at conducting electricity and heat. However, mercury is a type of metal that is in liquid form. Multiple types of metals are there, which remain in their solid state at room temperature and have a silver shine too. The metals are good conductors of electricity and heat, and they can also be converted into thin sheets and long wires as they are malleable and ductile. Metals, non-metals, and metalloids are different elements with different identities and properties.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
